|
TransCode has developed an RNA-based screening assay which has the potential to inform earlier therapeutic treatment along with clarity of response to therapy. Potentially it could serve as targeted companion diagnostic to the therapeutics that are being developed by TransCode because they can directly measure the abundance of the therapeutic target in the course of treatment. This could mean that patients could be stratified pre-treatment based on target biomarker expression and during treatment based on efficacy of target engagement. This approach represents a prime example of personalized medicine.
|
Predictive biomarker of metastasis
TransCode Therapeutics is continuing research and development on a specific biomarker assay (TCDx-miR-XXX) that has the potential to quantify microRNA expression in patient serum and blood. TransCode’s microRNA assay is designed to address a major unmet need in the areas of cancer biology, diagnosis, and therapy by introducing a tool that could permit the analysis of microRNA expression in blood.
- TransCode's predictive biomarker screening assay has the unique capability of microRNA profiling in blood samples.
- The fluorescent read-out generated by the assay is highly specific and has nanomolar sensitivity.
- The assay is inexpensive and rapid. It can be used to monitor treatment response with TTX-MC138 in clinical trials.
Click on image to enlarge
TransCode’s assay has the potential to address a major unmet need in the areas of cancer biology, diagnosis, and therapy by analyzing microRNA expression in blood.
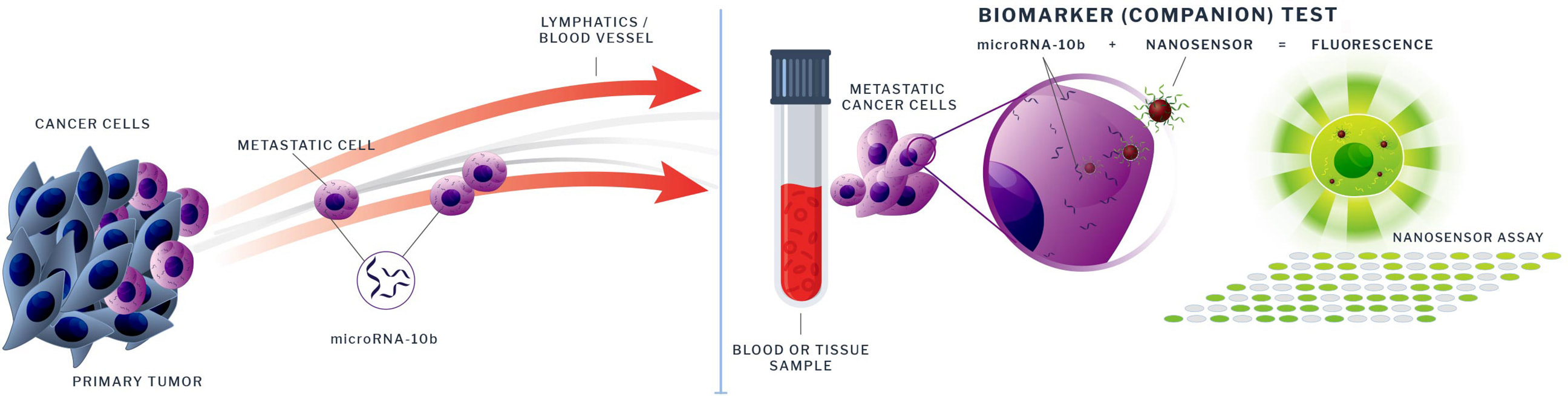
Potential utility of biomarker test for microRNA-10b
TransCode’s assay could address a major unmet need in the areas of cancer biology, diagnosis, and therapy by analyzing microRNA expression in blood.
- Early diagnosis has previously shown to reduce mortality in cancer patients.
- TransCode’s patented biomarker test is currently being optimized to measure the expression of microRNA-10b in patients.
- This test could provide unique information about cancer staging, disease progression, metastatic potential and ultimately survival, and could have an impact as a guide to therapy.
- Measuring microRNA-10b levels in the tumor before beginning treatment could better inform therapeutic decisions as evidenced in recent studies:
- MicroRNA-10b expression has been shown to be negatively correlated to sensitivity to 5-fluorouracil (5-FU)-based therapies;
- MicroRNA-10b expression has been shown to induce greater tamoxifen resistance;
- MicroRNA-10b expression has shown to discriminate between high-risk and low-risk disease with the potential to identify the presence of metastasis.
The FDA has not evaluated or approved the biomarker test for microRNA-10b and it is not currently available for patient use.